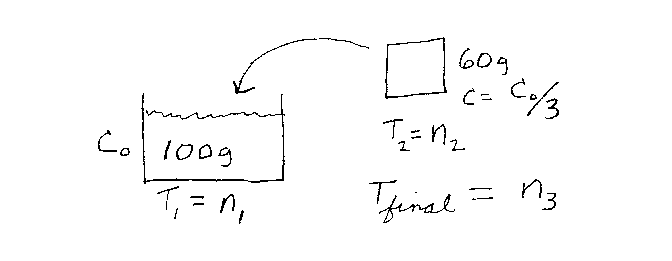
Problem D3:
You have 100 grams of a fluid that is initially at a temperature n1.
The specific heat capacity of the fluid is C0. You have 60 grams of
a solid that is initially at a temperature of n2. The specific heat
capacity of the solid is C0/3. You now place the solid into the
liquid and the system comes to thermal equilibrium at a temperature of
n3. What is n3?
Note that n1, n2, and n3 are all in the same
temperature units.
If you are currently in my class, you can record your grade by entering your name and student ID
number (without the leading zeros) below and clicking on "record grade".

